 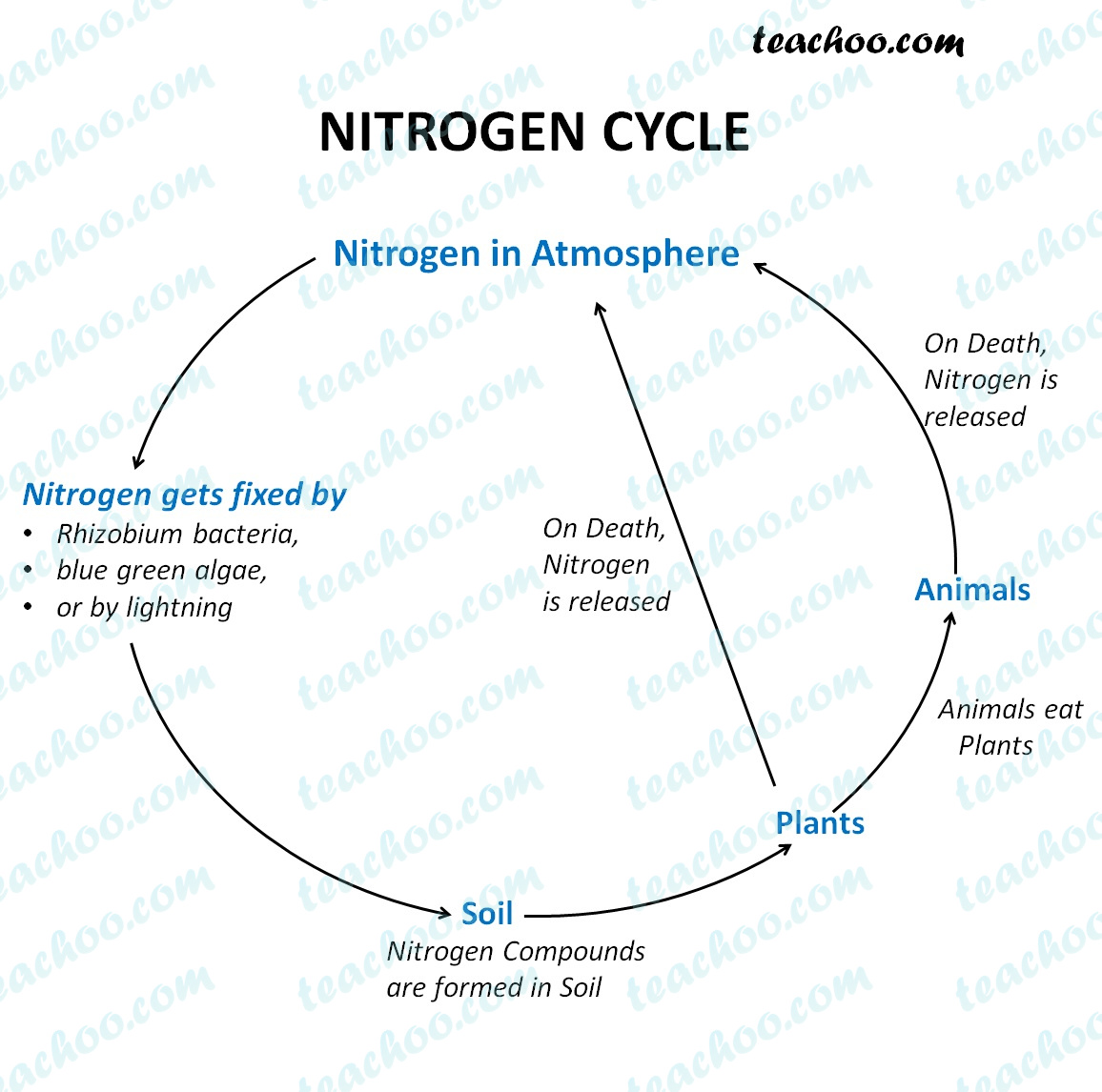
As the temperature increases, the entropy of the atoms in the lattice increase. Entropy is central to the second law of thermodynamics, which states that the entropy of an isolated system left to spontaneous evolution cannot decrease with time. Qualitatively, entropy is simply a measure how much the energy of atoms and molecules become more spread out in a process and can be defined in terms of statistical probabilities of a system or in terms of the other. The position of the atoms or molecules in the crystal would be perfectly defined. Entropy is a state function that is often erroneously referred to as the state of disorder of a system. Furthermore, their specific Gibbs free energies are the same, that is,Ġ = \Delta g (T,P) = g_$, meaning that the entropy decreasing with increasing pressure is equivalent to the volume increasing with increasing temperature. The entropy of a pure crystalline substance at absolute zero (i.e. When the two phases coexist, they have a common temperature $T$ and a common pressure $P$. When we say that the entropy of the gaseous phase is larger than that of the liquid phase, we are referring to the specific entropies (i.e., entropy per mole) of these phases when they coexist. Which of the following factors might make an endothermic reaction spontaneous An increase in entropy. Which state of matter has the lowest entropy Solid. 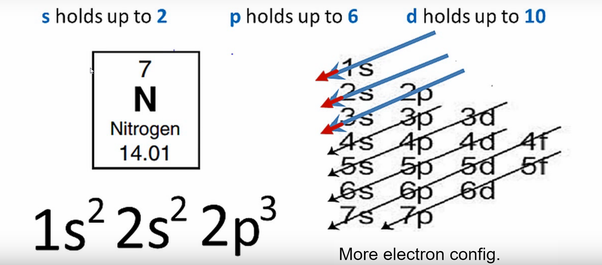
The free parameter in OP's example is the fraction of molecules in the liquid (or gaseous) phase. Terms in this set (5) Which state of matter has the highest entropy Gas. 
According to this law, a system with a fixed internal energy self-adjusts its free parameters in such a way that the entropy is maximized. Gases, because they are free to move about the entire volume of any container have much more. As OP noted, this is a consequence of the second law of thermodynamics. Which physical state of nitrogen has the highest entropy The gas phase. Whatever one observes (whether it is a liquid, a gas, or a mixture thereof) is the state of maximum entropy for the particular values of the internal energy and volume the system currently has. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
